This decommissioned ERA site remains active temporarily to support our final migration steps to https://ualberta.scholaris.ca, ERA's new home. All new collections and items, including Spring 2025 theses, are at that site. For assistance, please contact erahelp@ualberta.ca.
- 487 views
- 442 downloads
Candyfloss
-
- Author(s) / Creator(s)
-
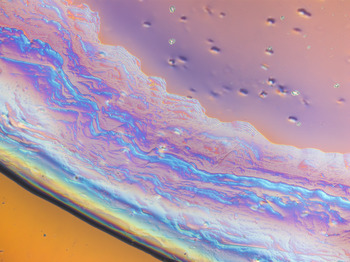
This image depicts a drop of sodium silicate (“liquid glass”) curing under polarized light. Sodium silicate, colloquially known as “waterglass” or “liquid glass”, can take the form of an aqueous solution or a solid glass. Liquid- form waterglass will solidify in contact with carbon dioxide; for this image, the carbon dioxide present in air was sufficient to initiate the solidification reaction.
Polarized light was used in this image to increase the visibility of the growing solid glass – the solid portion of this sample (the “waves”) is optically anisotropic (i.e. different areas of the sample interact differently with light), which resulted in the contrast between different thicknesses and orientations of the solid. Compare this to the liquid portion of the sample (upper right), which appears to be generally homogenous (optically isotropic).
Sodium silicate is being investigated as a candidate material for a bioactive glass composite that will be used for bone repair. This composite is designed to act as a scaffold for new bone growth and will dissolve once the patient’s bone has regrown. This particular image was used to investigate the solidification of sodium silicate in real time, to better understand and predict how it will behave in the aforementioned composite. // Program of Study: Master's // Faculty/Department: Faculty of Engineering, Department of Chemical and Materials Engineering // Place of creation: Chemical and Materials Engineering Building, University of Alberta // Award: Semi-finalist Prize, Images of Research Competition 2018 -
- Date created
- 2018-01-01
-
- Subjects / Keywords
-
- Type of Item
- Image
-
- License
- Attribution 4.0 International